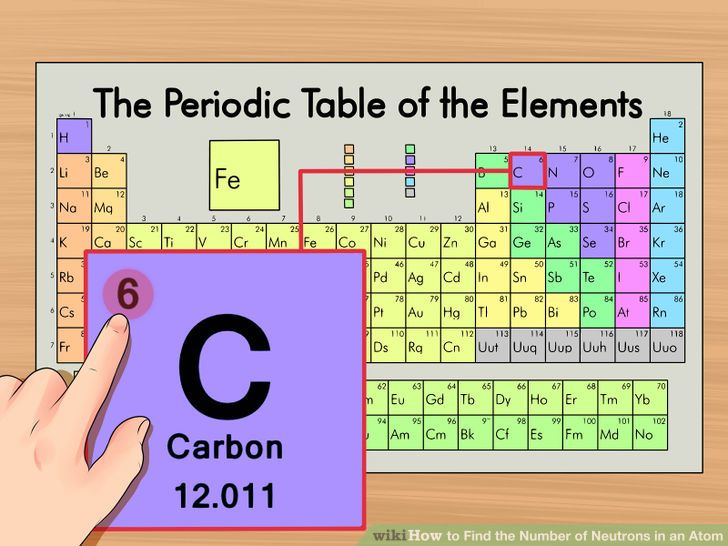
Something like that - is that you have a nucleus at theĬenter of an atom. Still show up at some government defense labs or These kind of nuclear drawings - and I'm sure that they Projects, you'll see a drawing that looks something And you might already beįamiliar with this if you look at old videos about atomic They all fit together and the structure of an atom. Made up of other more fundamental particles. Now we know that it actually isĬuttable and even though it is not a trivial thing, it's Or any element to that you encounter in the universe. And it doesn't just have to justīe for an apple they said this is true for any substance Some little part of an apple that they won't be able Said, well, at some point we think that there's going to be It's really just a mentalĪbstraction that allows us to describe a lot of observations 
Philosophical construct, which frankly, in a lot of ways, isn't It and they just felt that, oh, if I could just get my knifeĪ little bit sharper, I could cut it again and again. Philosophers went out there with a knife and tried to do 
So small, so tiny, that you can't cut it anymore. You have a nice looking apple,Īnd you just kept cutting it, smaller and smaller pieces. The apple - let me draw a nice looking apple just so If I started off with an apple, and I just kept cutting They said, hey, you know, if I started off with, I don't know, Philosophers long ago, and you could look it up on theĭifferent philosophers who first philosophized about it, The most philosophically interesting part of the whole Just starts right from the get-go with what's arguably Philosophically interesting things, but in chemistry it Pretty advanced before you start addressing the On the other hand, in a gas, there are no atomic bonds at all.The atoms do not want to stick together at all, and that forms a gas. In a fluid, there are atomic bonds, but they are just very weak. The atomic bonds don't form a hard surface to hold you up. Thus, if you attempted to "stand" on some fluid, you would sink right through LOL. The forces tend to "cancel out" more rather than form bonds. They tend not to want to borrow or give electrons to other atoms as much, so there bonds are weaker for that reason. The electromagnetic force is just a strong, but there is less room for bonds in those atoms. They are weak, and don't want to bind enough to hold you up. On the other hand, in a fluid, for example, the atomic bonds aren't quite so strong. When releasing the energy from just a small pellet of uranium in a radioactive way, a very destructive nuclear bomb occurs that can easily destroy entire cities, so that should give you a picture.) (Believe me, atoms hold ENOURMOUS amounts of energy and electromagnetic bonds between atoms can be very strong. Those hundreds of pounds are distributed throughout the whole floor onto all of those atoms. The atoms want to bind in that way, and the force is actually strong enough to hold hundreds of pounds easily. These fibers are held together by trillions of atomic bonds that are very strong. On a floor, for example, when you are standing on it, it is able to hold you up because there are trillions and trillions of atoms that form strong fibers in the wood. The atoms hold together to create a strong surface that appears to be solid. Your eyes aren't nearly sharp enough to see "holes" in the surface, and neither are nearly all microscopes. ) The forces are strong enough to make it feel solid, and the atoms have forces that are so strong that they don't like to collapse. (If you were to touch it on a computer screen, or on a piece of paper. Your nerves are feeling the surface of millions of atoms even in the period at the end of this sentence. Well, the forces in the atom are very strong, so even though it is mostly free space, the electromagnetic force keeps a piece of wood together. When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. 
As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
